Laboratory documentation is intended to ensure safety. It makes processes traceable, ensures quality, and facilitates audits. That's the theory, at least.
In practice, things look different: in many laboratories, documentation now takes up so much time that it feels like a second full-time job. Between logbooks, Excel lists, test equipment monitoring, calibration plans, and maintenance logs, there is hardly any room left for what really matters—clean analytics and scientific work.
The problem is not a lack of motivation. The problem is the system itself.
Documentation in the laboratory: many isolated solutions, no overall picture
Over the years, parallel systems have developed in most laboratories:
- Paper logbooks for device usage
- Excel spreadsheets for test equipment monitoring
- Separate maintenance lists in various formats
- Calibration logs in device folders
- QM documents on the network drive
- Manuals are everywhere—just never where you need them
Each element has its purpose. But together, they result in fragmented quality management in the laboratory—with noticeable consequences:
Typical problems in everyday documentation:
- Entries will be added later when there is time.
- Information is incomplete or contradictory
- Data exists twice in different systems
- Transcription errors from paper to Excel are becoming more frequent
- Audits cause stress because evidence has to be painstakingly gathered together.
This does not happen because of negligence. It happens because the system runs counter to the natural workflow.

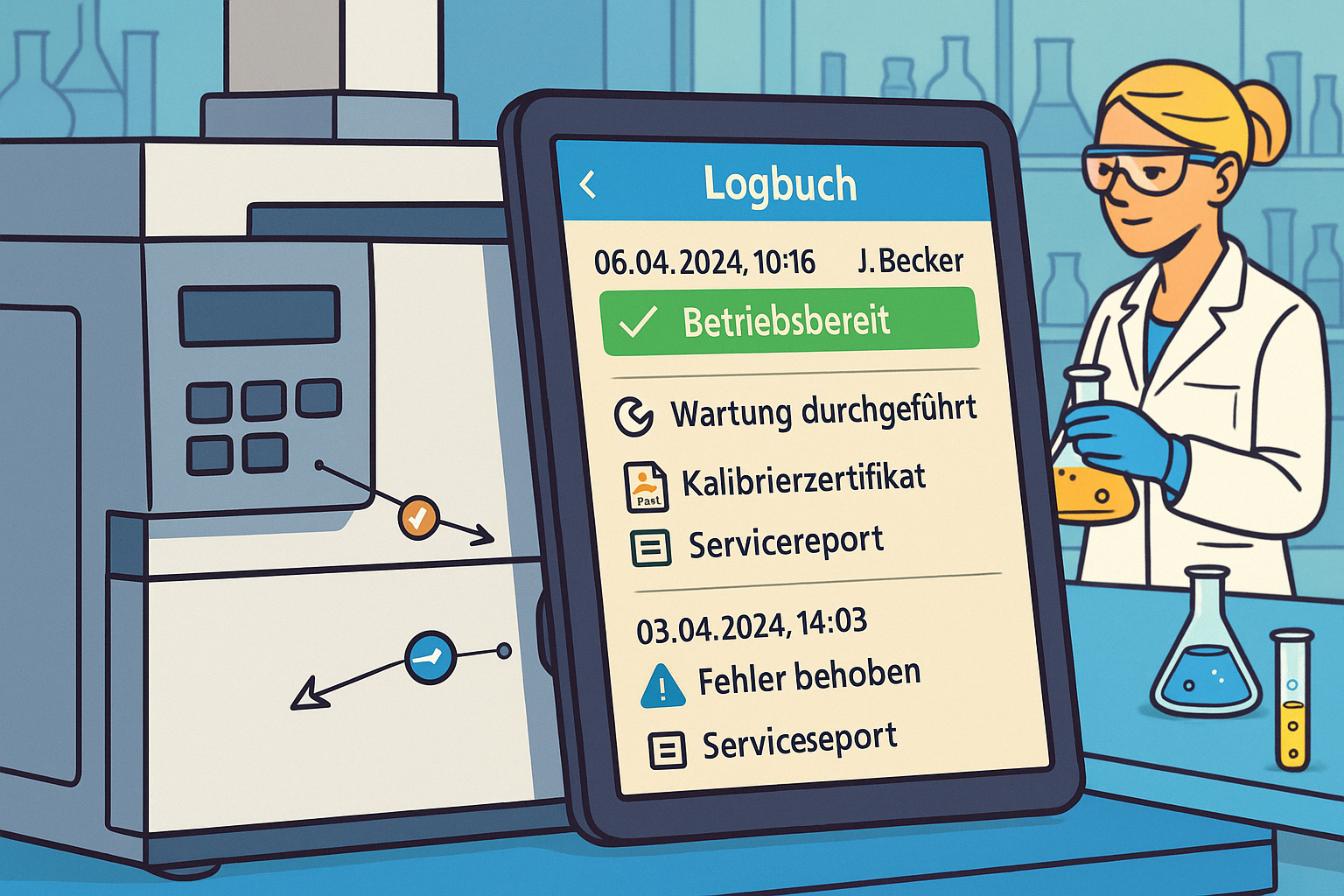
Excessive documentation costs time, concentration, and motivation in everyday laboratory work. A paperless laboratory provides a remedy by digitally linking processes, devices, and data. Modern solutions such as LabThunder to automate documentation, reduce errors, and significantly simplify the effort involved.
Paperless lab: Less friction, more clicks
Many associate digitization with the fear of "even more masks, even more mandatory fields, even more clicks."
A paperless laboratory should achieve exactly the opposite. When implemented correctly, digitization means:
- Information is recorded directly at the point of origin.
- The system automatically suggests recurring data.
- Equipment, maintenance, calibration, and usage are linked
- Documentation runs in the background—without any extra effort
The goal is not more documentation. The goal is less friction and better quality assurance in the laboratory.
Test equipment monitoring: Where Excel reaches its limits
Test equipment monitoring is a critical issue in many laboratories. It is often still organized via:
- Excel lists (test equipment monitoring Excel is standard)
- Manual calendar reminders
- Handwritten checklists
- Email chains between colleagues
These methods work—until they don't.
Typical risks associated with manual test equipment monitoring:
- Calibration dates are overlooked
- Responsibilities are unclear or outdated
- The status of individual devices is not transparent
- Audits lack complete documentation
- Locked devices are still being used
This is precisely where seamless quality assurance in the laboratory is crucial. An overlooked or expired test device can not only call individual measurements into question—in the worst case, it can jeopardize entire batches or approvals.
Test equipment monitoring and calibration: These must go hand in hand
Test equipment monitoring calibration is not an isolated process. It is directly related to equipment usage:
- When was the device last used?
- Is the calibration still valid?
- Who is responsible for implementation?
- What evidence already exists?
If this information is scattered, gaps arise. And gaps mean risk.
Usability determines data quality
A system can be technically perfect—if it feels complicated, it will be avoided in everyday use.
Then the following arise:
- Addenda at the end of the week
- Abbreviations that are "actually prohibited"
- Entries with "Let's do it later"
- Excel shadow lists alongside the official system
Good quality management laboratories therefore need one thing above all else: user-friendliness.
Software must be designed in such a way that it:
- understands the actual workflow
- is logically structured
- automatically supported instead of blocked
- faster than the Excel alternative
Only then does documentation become part of the routine—instead of a tedious additional task.
How modern laboratory systems simplify documentation
Modern software for paperless laboratories connects all relevant areas:
- Devices and their master data
- Maintenance and calibration history
- Logbooks with usage data
- Test equipment monitoring with automatic reminders
- Audit-ready reports at the touch of a button
Practical examples from everyday laboratory work:
- A device is used → The use is automatically documented
- A calibration date is approaching → The system reminds the person responsible
- A test device is locked → Use is no longer technically possible
- An audit is coming up → All evidence is available digitally and in full
This means that documentation is not "processed" retrospectively. It happens during normal work—in the background, without any additional effort.
Why the issue is becoming more urgent now
Laboratories today operate under different conditions than they did ten years ago:
- Regulatory requirements are constantly increasing
- Audits are becoming more detailed and frequent
- Documentation requirements are increasing
- At the same time, staffing levels often remain the same.
More manual documentation is not a solution under these conditions. It exacerbates the problem.
The answer lies in:
- Integration instead of isolated solutions —one system instead of ten lists
- Automation instead of addenda – documentation during the process
- Clear responsibilities – who does what by when
- Software that supports people —not burdens them
Conclusion: Rethinking documentation
The problem is not documentation itself. The problem is how it is often organized today.
When test equipment monitoring, calibration, maintenance, and usage come together in a single, easy-to-use system, perceptions change fundamentally.
Documentation no longer feels like control. It becomes what it should be: security and relief.



LabThunder:
✅ Compliant with ISO 17025, GMP/GLP, and ISO 15189
✅ Digital logbooks instead of paper chaos
✅ Thunder AI central intelligence for errors & questions
✅ Smart & predictive maintenance prevents downtime
✅ Greater independence from external service providers
✅ Up to 50% fewer service calls
✅ Easy to use - no IT required
Contact us today for a free demo:





%20new.png)
















.jpg)
.jpg)

